Many animals use dissolved chemical cues (or odors) to find food, mates, and avoid predators. Crustaceans (including crabs and lobsters) and insects accomplish this task with external `noses:’ antennae studded with chemosensory hairs. Crabs waive these antennae through the water (or air) to collect odor molecules from the surrounding fluid.
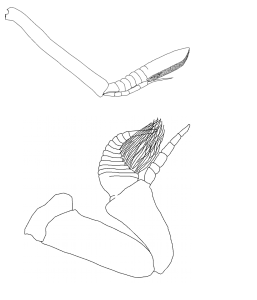
The arrangement, number, and size of the chemosensory hairs, as well as how fast they are moved, determine the amount of odor that the animal can capture. We use a variety of techniques to explore how these arrays of hairs capture odor in air and water and how they could have evolved.
Particle image velocimetry is used to quantify flow fields on dynamically scaled physical models of chemosensory hair arrays. PIV uses the motion of reflective particles in a 2D plane to reconstruct velocity vector fields within the array.
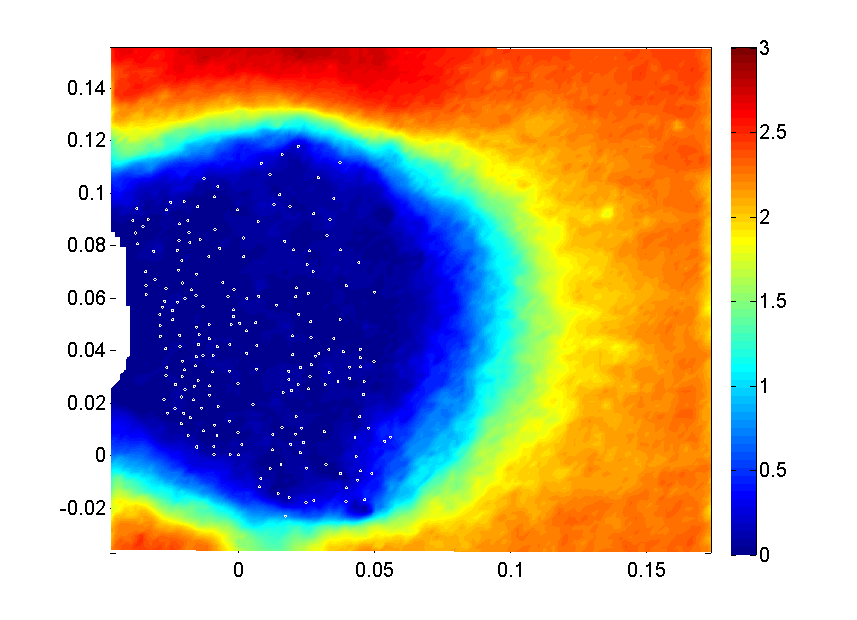
In collaboration with several mathematicians (Dr. Shilpa Khatri, Univ. of California, Merced; Dr. Laura Miller, Univ. of North Carolina at Chapel Hill; Dr. Yanyan He, New Mexico Tech), computational simulations can explore odor capture beyond what’s possible with experimentation. Below is an example odor capture simulation of a odor filament in water being captured by a marine blue crab array. The advection-diffusion model was by Dr. Shilpa Khatri using PIV velocity data (from Waldrop et al. 2016).
Current work includes simulating fluid flow through a variety of hair-array morphologies with the immersed boundary method and marrying these velocity fields with diffusion model and uncertainty quantification (with Dr. Yanyan He, New Mexico Tech) to explore how variation affects the odor-capture performance of hair arrays.
Associated Publications
L.D. Waldrop, Y. He, S. Khatri. 2018. What can computational modeling tell us about the diversity of odor-capture structures in the Pancrustacea? Journal of Chemical Ecology https://link.springer.com/article/10.1007/s10886-018-1017-2. Full text.
L.D. Waldrop, L.A. Miller, S. Khatri. 2016. A tale of two antennules: The performance of crab odor-capture organs in air and water. Journal of the Royal Society Interface 13: 20160615. DOI: http://dx.doi.org/10.1098/rsif.2016.0615. Full text.
L.D. Waldrop, M.A.R. Koehl. Do terrestrial hermit crabs sniff? Air flow and odorant capture by flicking antennules. 2016. Journal of the Royal Society Interface 13(114): 20150850. DOI: 10.1098/rsif.2015.0850. Full text.
L.D. Waldrop, M.A. Reidenbach, M.A.R. Koehl. 2015. Flexibility of crab chemosensory hairs enables flicking antennules to sniff. Biological Bulletin 229(2): 185-198. Full text.
L.D. Waldrop, M. Hann*, A. Henry*, A. Kim*, A. Punjabi*, M.A.R. Koehl. 2015. Ontogenetic changes in the olfactory antennules of the shore crab, Hemigrapsus oregonensis, maintain sniffing function during growth. Journal of the Royal Society Interface 12: 20141077. Full text.
L.D. Waldrop, R.M. Bantay*, Q.V. Nguyen*. 2014. Scaling of olfactory antennae of the ter- restrial hermit crabs Coenobita rugosus and Coenobita perlatus during ontogeny. PeerJ 2:e535. Full text.
L.D. Waldrop. 2013. Ontogenetic scaling of the olfactory antennae and flicking behavior of the shore crab, Hemigrapsus oregonensis. Chemical Senses 38(6): 541-550. Full text.
All publications available on request.